 Tracking Side Effects Once a Vaccine is Administered To learn about FDA’s role in the vaccine approval process, consult FDA’s Vaccine Product Approval Process web page. FDA can require each manufacturer submit samples of each vaccine lot for testing. Monitoring of the vaccine and of production activities, including periodic facility inspections, must continue as long as the manufacturer holds a license for the vaccine product.įDA can require a manufacturer submit the results of their own tests for potency, safety, and purity for each vaccine lot. Presentation of findings to FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC)Īfter approving a vaccine, FDA continues to oversee its production to ensure continuing safety.Inspection of the manufacturing facility.An Investigational New Drug application.The sponsor of a new vaccine product follows a multi-step approval process, which typically includes 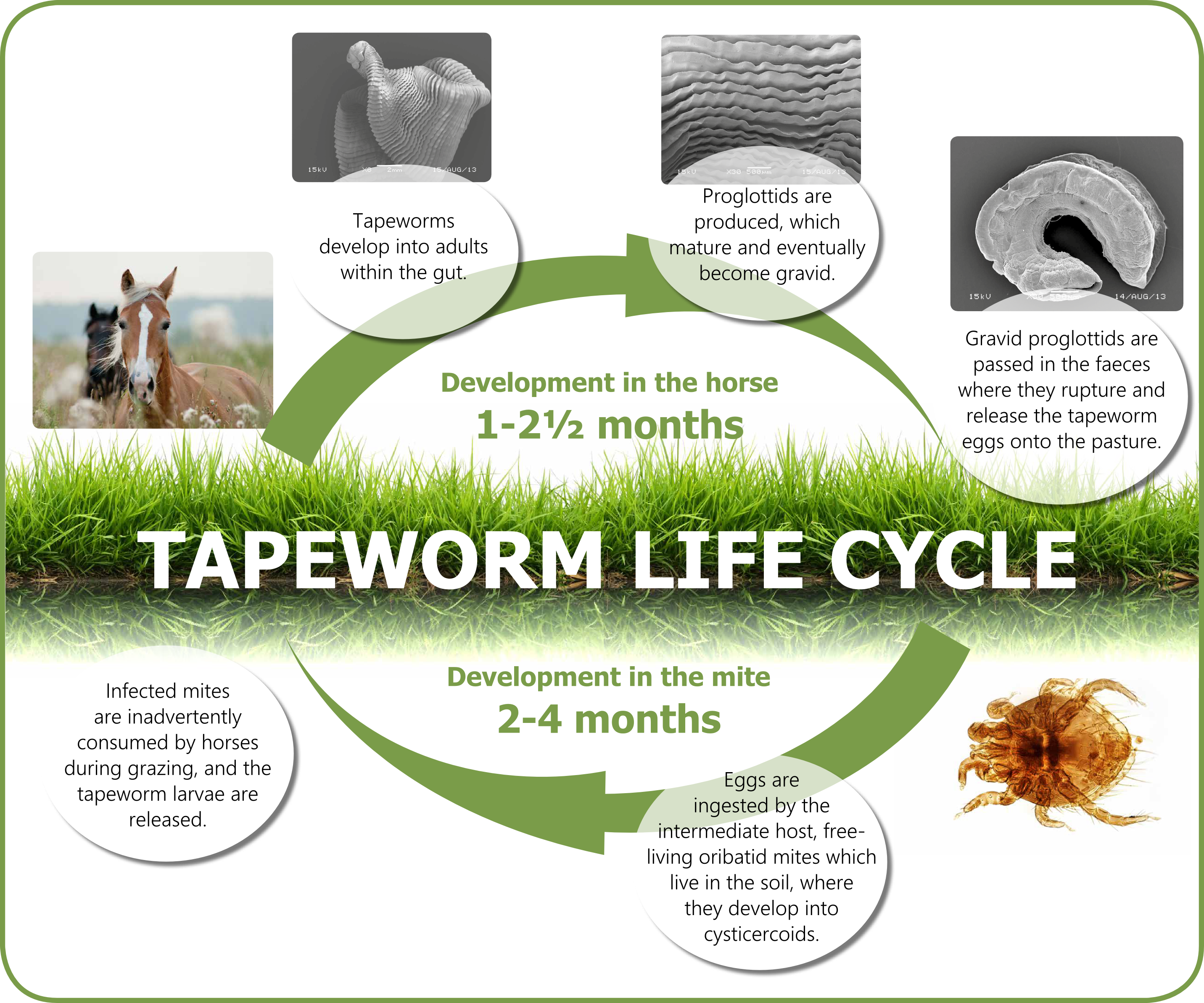
Food and Drug Administration’s (FDA’s) Center for Biologics Evaluation and Research (CBER) is responsible for regulating vaccines in the United States. See how a new vaccine is developed, approved, manufactured, added to recommended schedule, and is continually monitored. Many vaccines undergo Phase IV formal, ongoing studies after the vaccine is approved and licensed.įor more information and to find out about new vaccines on the horizon, see the World Health Organization’s (WHO’s) Development of New Vaccines web page. In Phase III, the vaccine is given to thousands of people and tested for efficacy and safety. In Phase II, the clinical study is expanded and vaccine is given to people who have characteristics (such as age and physical health) similar to those for whom the new vaccine is intended. During Phase I, small groups of people receive the trial vaccine. 
The general stages of the development cycle of a vaccine are:Ĭlinical development is a three-phase process. This page leads to other pages that describe vaccine development and testing such as basic research, clinical studies, side effects and adverse reactions, vaccines of the future, and the vaccine product approval process. Tracking Side Effects Once a Vaccine is Adminstered. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2022
Categories |
 RSS Feed
RSS Feed
